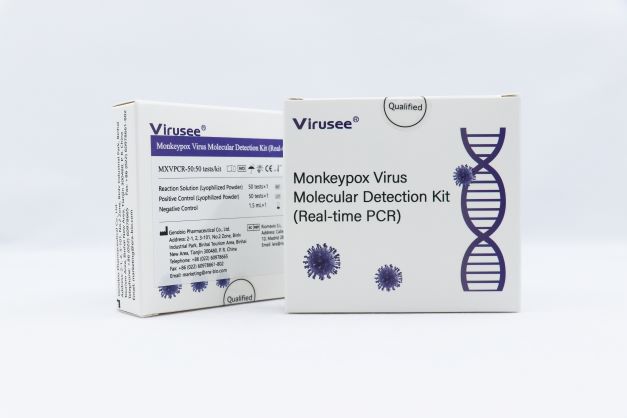
Reliable Supplier China CE Approved Monkeypox Virus Extraction Test Kit
With our wealthy encounter and considerate services, we now have been recognized for a trusted provider for most worldwide customers for Reliable Supplier China CE Approved Monkeypox Virus Extraction Test Kit, We now have ISO 9001 Certification and qualified this product .over 16 years experiences in manufacturing and designing, so our products and solutions featured with ideal top quality and aggressive value. Welcome cooperation with us!
With our wealthy encounter and considerate services, we now have been recognized for a trusted provider for most worldwide customers for China Rapid Kit, in Vitro Diagnostic Reagents, With the goal of “zero defect”. To care for the environment, and social returns, care employee social responsibility as own duty. We welcome friends from all over the world to visit and guide us so that we can achieve the win-win goal together.
Product Introduction
Transport under room temperature!
Virusee® Monkeypox Virus Molecular Detection Kit (Real-time PCR) is used for in vitro quantitative detection of F3L gene from Monkeypox virus in skin lesions, vesicles and pustular fluid, dry crusts and other specimens from individuals who are suspected of Monkeypox virus infection by their healthcare provider.
The product can be transported under room temperature, stable and reduces costs.
Characteristics
|
Name |
Monkeypox Virus Molecular Detection Kit (Real-time PCR) |
|
Method |
Real-time PCR |
|
Sample type |
Skin lesions, vesicles and pustular fluid, dry crusts, etc. |
|
Specification |
25 tests/kit, 50 tests/kit |
|
Detection time |
1 h |
|
Detection objects |
Monkeypox virus |
|
Stability |
The kit is stable for 12 months at 2°C-8°C in dark |
|
Transportation conditions |
≤37°C, stable for 2 months |
|
Inter assay variation |
≤ 5% |
|
Limit of Detection |
500 copies/mL |
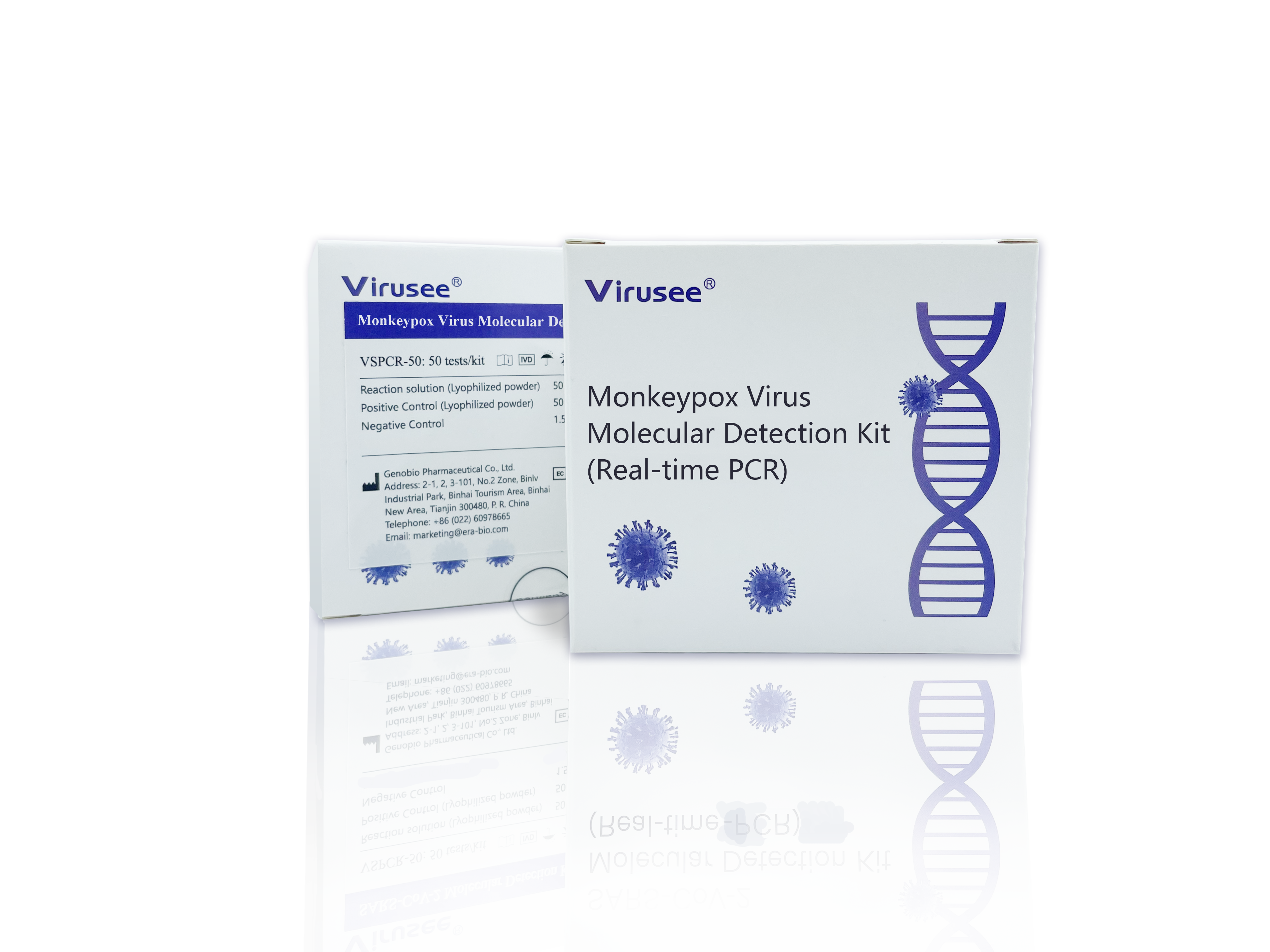
Advantage
- Accurate
High sensitivity and specificity, qualitative results
Strictly controls the experiment quality with positive and negative controls - Economic
The reagents are in terms of lyophilized powder, reducing storage difficulty.
The kit can be transported at room temperature, minimizing transportation cost.
- Flexible
Two specifications available. Users can choose between 25 T/Kit and 50 T/Kit
What is Monkeypox Virus?
Monkeypox is a viral zoonosis (a virus transmitted to humans from animals) with symptoms similar to those seen in the past in smallpox patients, although it is clinically less severe. With the eradication of smallpox in 1980 and subsequent cessation of smallpox vaccination, monkeypox has emerged as the most important orthopoxvirus for public health. Monkeypox primarily occurs in central and west Africa, often in proximity to tropical rainforests, and has been increasingly appearing in urban areas. Animal hosts include a range of rodents and non-human primates.
Transmission
Animal-to-human (zoonotic) transmission can occur from direct contact with the blood, bodily fluids, or cutaneous or mucosal lesions of infected animals. In Africa, evidence of monkeypox virus infection has been found in many animals including rope squirrels, tree squirrels, Gambian pouched rats, dormice, different species of monkeys and others. The natural reservoir of monkeypox has not yet been identified, though rodents are the most likely. Eating inadequately cooked meat and other animal products of infected animals is a possible risk factor. People living in or near forested areas may have indirect or low-level exposure to infected animals.
Human-to-human transmission can result from close contact with respiratory secretions, skin lesions of an infected person or recently contaminated objects. Transmission via droplet respiratory particles usually requires prolonged face-to-face contact, which puts health workers, household members and other close contacts of active cases at greater risk. However, the longest documented chain of transmission in a community has risen in recent years from 6 to 9 successive person-to-person infections. This may reflect declining immunity in all communities due to cessation of smallpox vaccination. Transmission can also occur via the placenta from mother to fetus (which can lead to congenital monkeypox) or during close contact during and after birth. While close physical contact is a well-known risk factor for transmission, it is unclear at this time if monkeypox can be transmitted specifically through sexual transmission routes. Studies are needed to better understand this risk.
Diagnosis
The clinical differential diagnosis that must be considered includes other rash illnesses, such as chickenpox, measles, bacterial skin infections, scabies, syphilis, and medication-associated allergies. Lymphadenopathy during the prodromal stage of illness can be a clinical feature to distinguish monkeypox from chickenpox or smallpox.
If monkeypox is suspected, health workers should collect an appropriate sample and have it transported safely to a laboratory with appropriate capability. Confirmation of monkeypox depends on the type and quality of the specimen and the type of laboratory test. Thus, specimens should be packaged and shipped in accordance with national and international requirements. Polymerase chain reaction (PCR) is the preferred laboratory test given its accuracy and sensitivity. For this, optimal diagnostic samples for monkeypox are from skin lesions – the roof or fluid from vesicles and pustules, and dry crusts. Where feasible, biopsy is an option. Lesion samples must be stored in a dry, sterile tube (no viral transport media) and kept cold. PCR blood tests are usually inconclusive because of the short duration of viremia relative to the timing of specimen collection after symptoms begin and should not be routinely collected from patients.
Reference: https://www.who.int/news-room/fact-sheets/detail/monkeypox
Order Information
|
Model |
Description |
Product code |
|
MXVPCR-25 |
25 tests/kit |
MXVPCR-25 |
|
MXVPCR-50 |
50 tests/kit |
MXVPCR-50 |
With our wealthy encounter and considerate services, we now have been recognized for a trusted provider for most worldwide customers for Reliable Supplier China CE Approved Monkeypox Virus Extraction Test Kit, We now have ISO 9001 Certification and qualified this product .over 16 years experiences in manufacturing and designing, so our products and solutions featured with ideal top quality and aggressive value. Welcome cooperation with us!
Reliable Supplier China Rapid Kit, in Vitro Diagnostic Reagents, With the goal of “zero defect”. To care for the environment, and social returns, care employee social responsibility as own duty. We welcome friends from all over the world to visit and guide us so that we can achieve the win-win goal together.